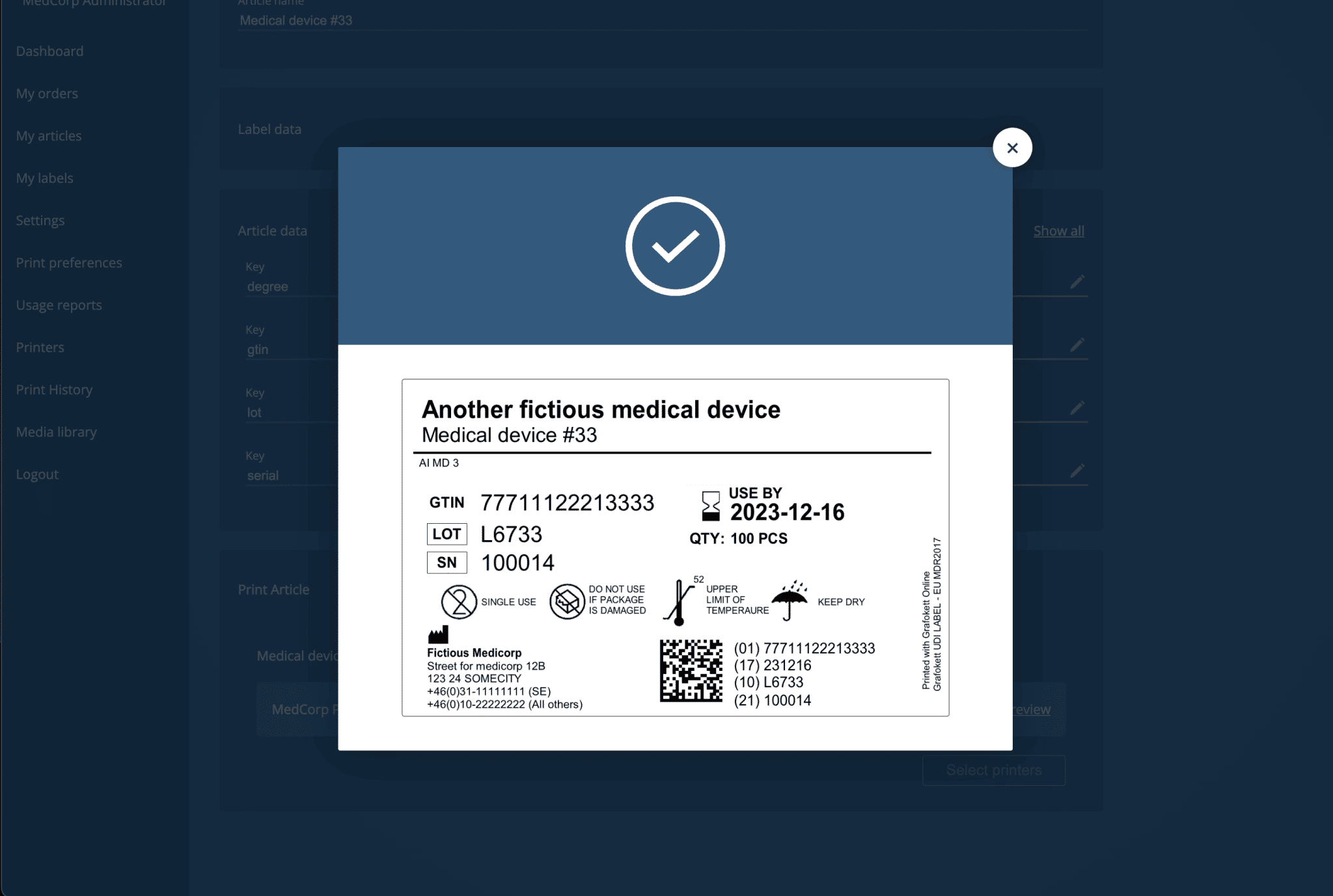
What is Basic UDI?
A registration number that identifies a group of medical devices with the same purpose, risk class and comparable design and manufacturing functions. In general, a Basic UDI-DI consists of four elements:
- A specific start for an issuing body (eg GS1 codes start with “01”)
- Manufacturer code
- Product group code
- Check digit
Basic UDI-DI is used to register the units in EUDAMED and must be referred to in relevant documents (ie in the certificates and technical documentation including operating instructions). However, it must not be stated either on the product packaging or on the product itself. The manufacturers of medical devices are responsible for maintaining a mapping between the Basic UDI-DI and all associated (underlying) UDI-DIs.
What is Basic UDI?
Basic UDI (Unique Device Identification) is a system introduced by the European Union’s Medical Device Regulation (MDR) 2017 that requires medical device manufacturers to assign unique identifiers to their products. The main purpose of the UDI system is to improve the traceability of medical devices and to enhance patient safety.
The MDR 2017 defines UDI as “a unique identifier assigned to a device, consisting of a device identifier (DI) and a production identifier (PI)”. The DI is a globally unique code that identifies the specific device model and its intended use, while the PI identifies the production information, such as the production date, the manufacturer’s identifier, and the batch number. The UDI system ensures that every medical device has a unique identifier that can be easily traced and accessed throughout its entire life cycle.
One of the key benefits of the UDI system is that it enables health authorities and healthcare providers to quickly retrieve information about a medical device in case of adverse events, incidents, or product recalls. This information includes the device’s history, usage, and any previous adverse events, which can help in the investigation and resolution of the issue.
In addition, the UDI system makes it easier for healthcare providers to keep track of the medical devices they use and to manage their inventory. This leads to increased efficiency and cost savings, as healthcare providers can avoid purchasing duplicates or expired devices.
To comply with the MDR 2017, medical device manufacturers must follow the EU UDI guidelines and must include the UDI on the device label and in the accompanying documentation. The UDI must also be entered into the European Databank on Medical Devices (EUDAMED) database, which is maintained by the European Commission.
In conclusion, the UDI system introduced by the MDR 2017 plays a crucial role in improving the safety and traceability of medical devices. It ensures that medical devices are identifiable, easily traceable, and that relevant information is readily available in case of adverse events, incidents, or product recalls. As a result, patients and healthcare providers can have confidence in the safety and performance of the medical devices they use.
Read all about it here UDI – Unique Device Identifier